Companion Diagnostics
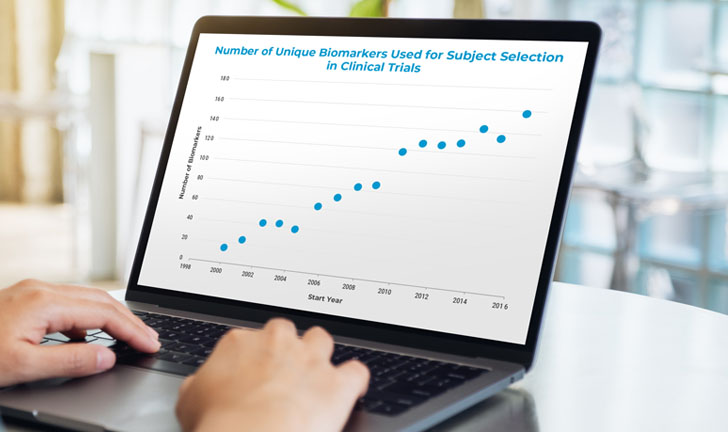
Biomarkers that reveal response or non-response to a particular biomarker or drug are at the center of the personalized medicine revolution. They are also becoming a growing requirement in clinical trials to allow for better patient stratification and outcome. For a company developing a therapy, having a biomarker strategy has become a necessity.
Flagship Biosciences has developed expertise in developing assays in the fields of oncology, immune-oncology, neuromuscular disorders and cell and gene therapies for rare diseases. Our experts are skilled in selecting antibodies, developing scoring schemes, setting cut-off points and developing testing strategies. Our services include:

- Determine the best commercially-available and novel/proprietary primary antibodies for targets of interest
- Select appropriate controls and test tissues
- Optimized dual DNA/RNA extraction services
- Genomic biomarker analysis (RNA sequencing, whole genome sequencing, exome sequencing, qPCR)
- Immunophenotyping and functional analysis via flow cytometry and cell-based services
- Experienced and proven, proprietary assay development approaches for IHC, ISH and multiplex IF
- Titration of primary antibody concentration and selection of optimal assays and detection systems
- Orthogonal testing via RNAScope, PCR, ELISA, Nanostring, etc.
- Consulting vis-à-vis diagnostic-specific regulatory requirements.
- Validation from RUO to LDT according to GMP
- Sensitivity, specificity and reproducibility testing
- Scoring scheme development driven by board-certified anatomical pathologists
- Expression rate analysis for cut-off determination
IVD Assays
The assays and details provided in the lists are not all-inclusive of Flagship’s experience. If the specific assay you are interested in is not listed, please contact us as we may have experience with the assay or can custom develop an assay to address your specific requirements.
TissueInsight®
For Rapid And Precise Cancer Care, Pathologists And Oncologists Need Dependable Diagnostic Testing To Connect Patients To Individualized Immuno-Oncology Therapies.
While tools exist for effectively managing slide images and streamlining workflow for digital pathology, little has been done to relieve the time-intensive burden of the creation, validation, and maintenance of image analysis tools and diagnostic algorithms. TissueInsight® from Flagship Biosciences puts the power of digital pathology in the hands of any pathologist at the touch of a button.